MicroBase Technology Corp. EU MDR Certified
(Source: MicroBase Technology)
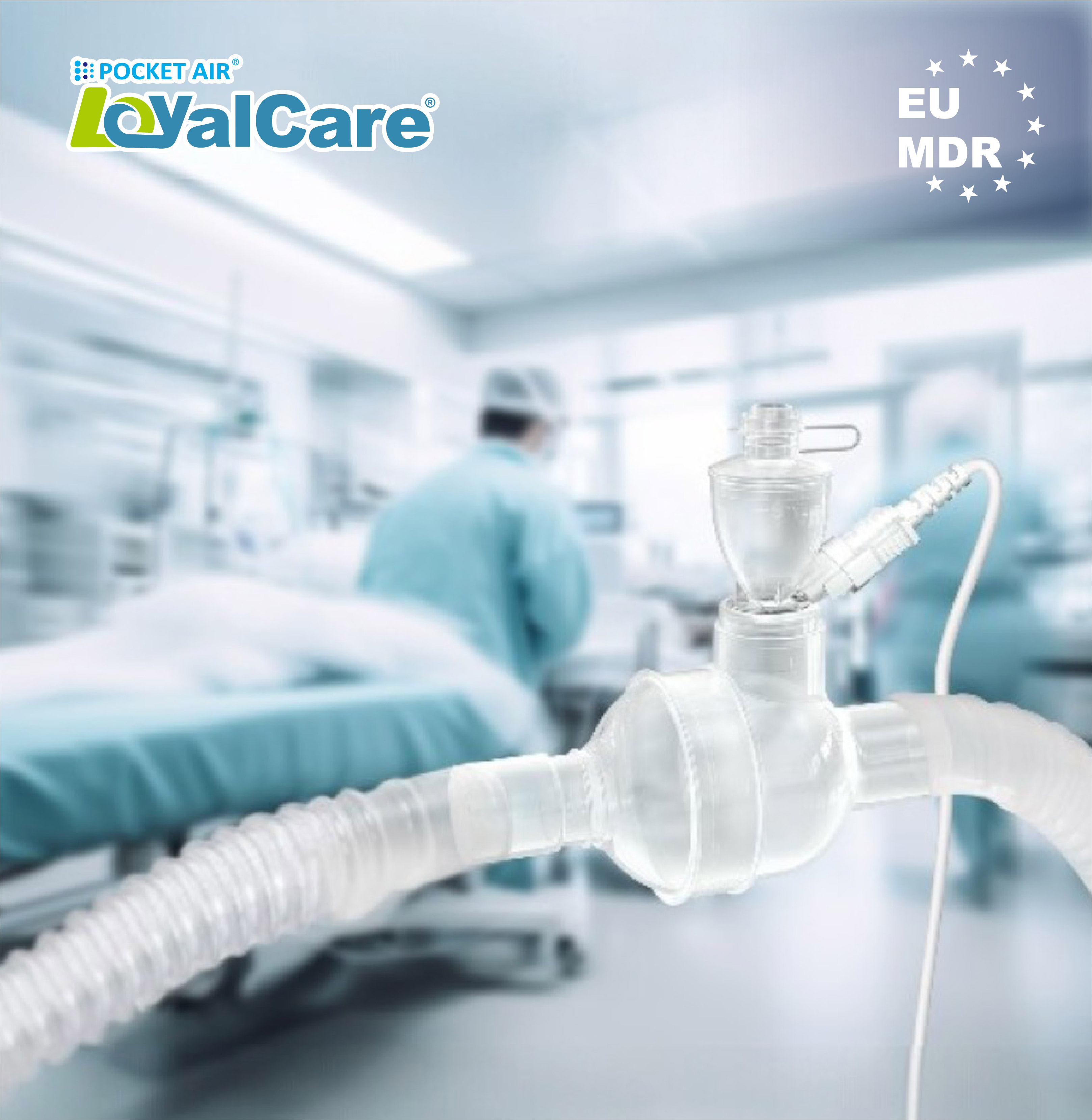
MicroBase Technology Corp. (TWSE: 3184), a leading innovator in vibrating mesh and aerosol drug delivery technologies, announced that its PocketAir® product portfolio has officially obtained EU Medical Device Regulation (MDR) certification, covering both its hands-free nebulizer for homecare and its inline nebulizer for ICU.
Unlocking the EU Market: MDR as a Strategic Time-to-Market Advantage
The EU MDR represents the most stringent regulatory framework for medical devices worldwide. Early compliance positions MicroBase as a first mover at a critical regulatory inflection point.
According to the transitional provisions of Regulation (EU) 2024/18601 , registration in the EUDAMED (European Database on Medical Devices) will become mandatory starting May 28, 2026. MicroBase’s portfolio is already fully compliant, allowing partners to bypass regulatory bottlenecks and accelerate market entry.
Dual Market Strategy: Precision Solutions for Homecare & Hospital
We address the full spectrum of chronic respiratory disease management (Asthma / COPD) through two complementary products:
USD 4.1 Billion Market Opportunity in Respiratory Care
According to World Health Organization (WHO)2 , more than 81.7 million patients in Europe are affected by chronic respiratory diseases. 2025 Research Nester report3 further projects the global vibrating mesh nebulizer market to reach USD 4.1 billion by 2037, with a 9.5% CAGR, underscoring strong long term demand for aerosol drug delivery technologies.
From Zero to MDR Approval
Achieving EU MDR, the newest and most demanding medical device regulation was accomplished through team work across regulation affairs, R&D and business development staffs. The dedicated task force synthesized over 1,000 clinical literatures to meet rigorous safety and efficacy standards.
With a track record of certifications and registrations in over 20 countries, we has established a scalable model for regulatory compliance and cross departmental collaboration.
This certification validates our integrated expertise in manufacturing and clinical evidence. We are now uniquely positioned as a key technology supplier in personalized medicine and precision drug delivery across the international stage.
3. Research Nester https://www.researchnester.com/tw/reports/vibrator-mesh-nebulizer-market/2913。