
Inline Aerosol Drug Delivery Solution
The world's first Polyimide (PI)-based closed aerosol drug delivery platform. Specially engineered for patients requiring mechanical ventilators or other positive pressure/high-flow respiratory support circuits, its expanded mist reservoir design significantly enhances lung deposition rates.
POLYIMIDE (PI) Advantages
During nebulization, aerosol particles are highly susceptible to electrostatic attraction, causing them to adhere to metal surfaces. Utilizing PI material minimizes static generation and reduces drug residue on circuit surfaces, thereby maximizing aerosol output and inhalation efficiency.


Shear Stress Control
Polyimide (PI) undergoes elastic deformation under fluid pressure, mitigating abrupt flow velocity changes and reducing shear rates. Unlike rigid metals, it prevents damage caused by amplified instantaneous stress.
Thermal Damage Prevention
PI’s low thermal conductivity ensures that heat does not accumulate on the working surface. This prevents localized hot spots, significantly reducing the risk of damaging sensitive bio-substances.
Reduced Surface Adsorption
With higher hydrophobicity than metals, PI minimizes protein and lipid adsorption, thereby inhibiting the degradation caused by interfacial shear stress.
Superior Chemical Stability
PI is chemically inert and non-metallic. It does not release metal ions during nebulization, ensuring that the properties of biological molecules or liposomes remain intact.
Advanced Micro-machining
The high processability of PI allows for the fabrication of precise microporous structures, ensuring product consistency and exceptional performance for high-accuracy nebulization needs.
Versatile Respiratory Therapy Support

Invasive Mechanical Ventilation
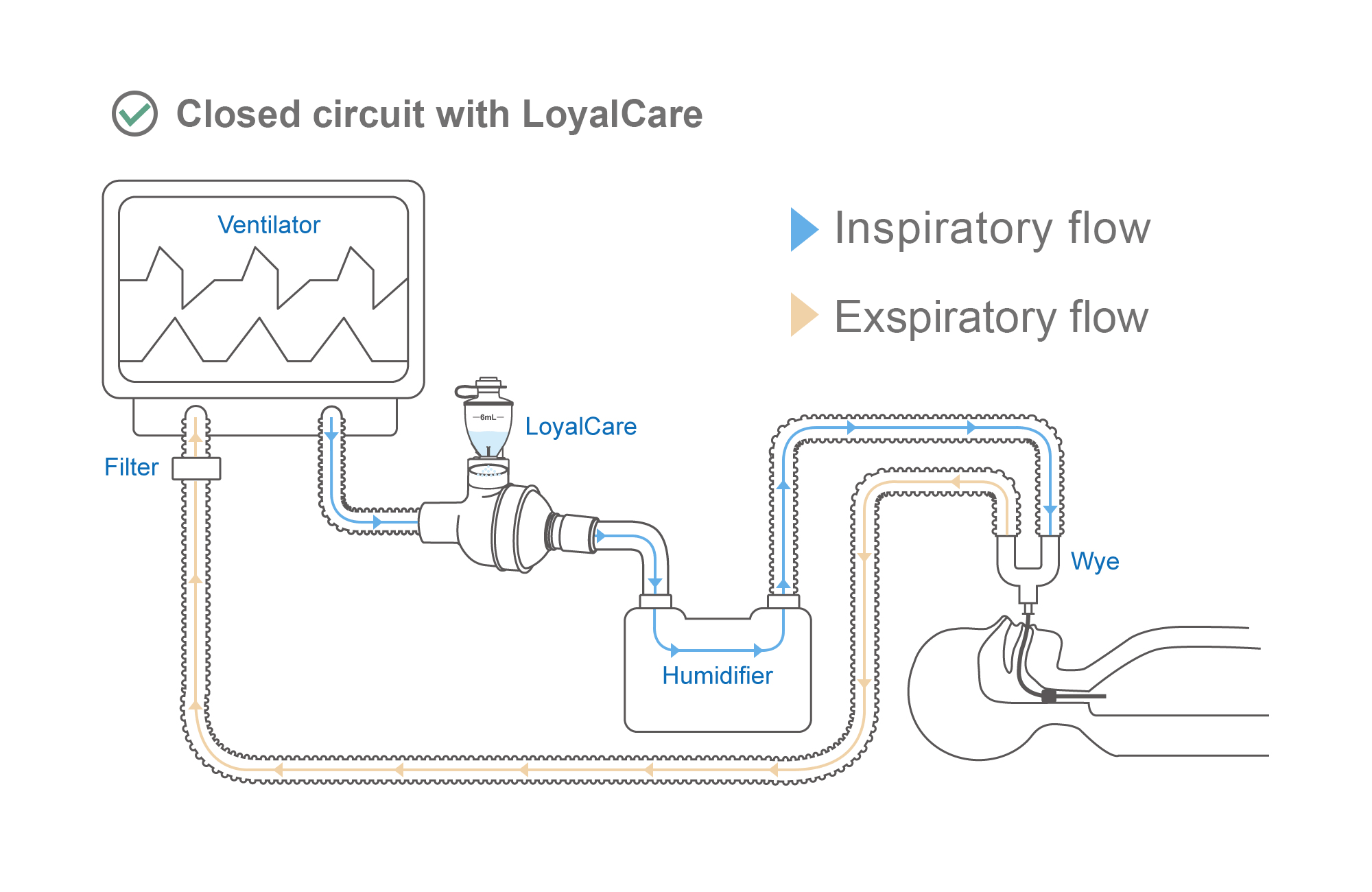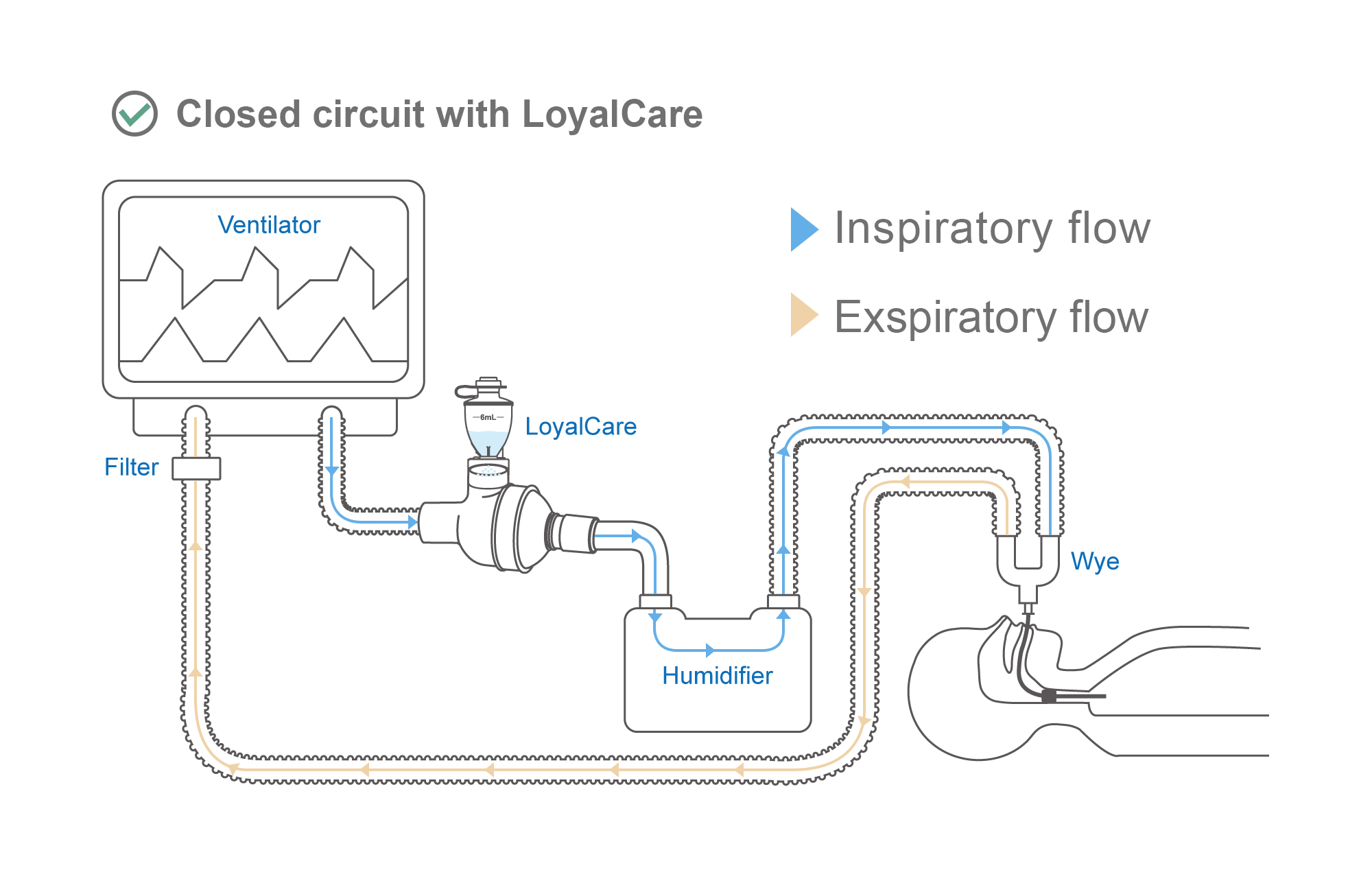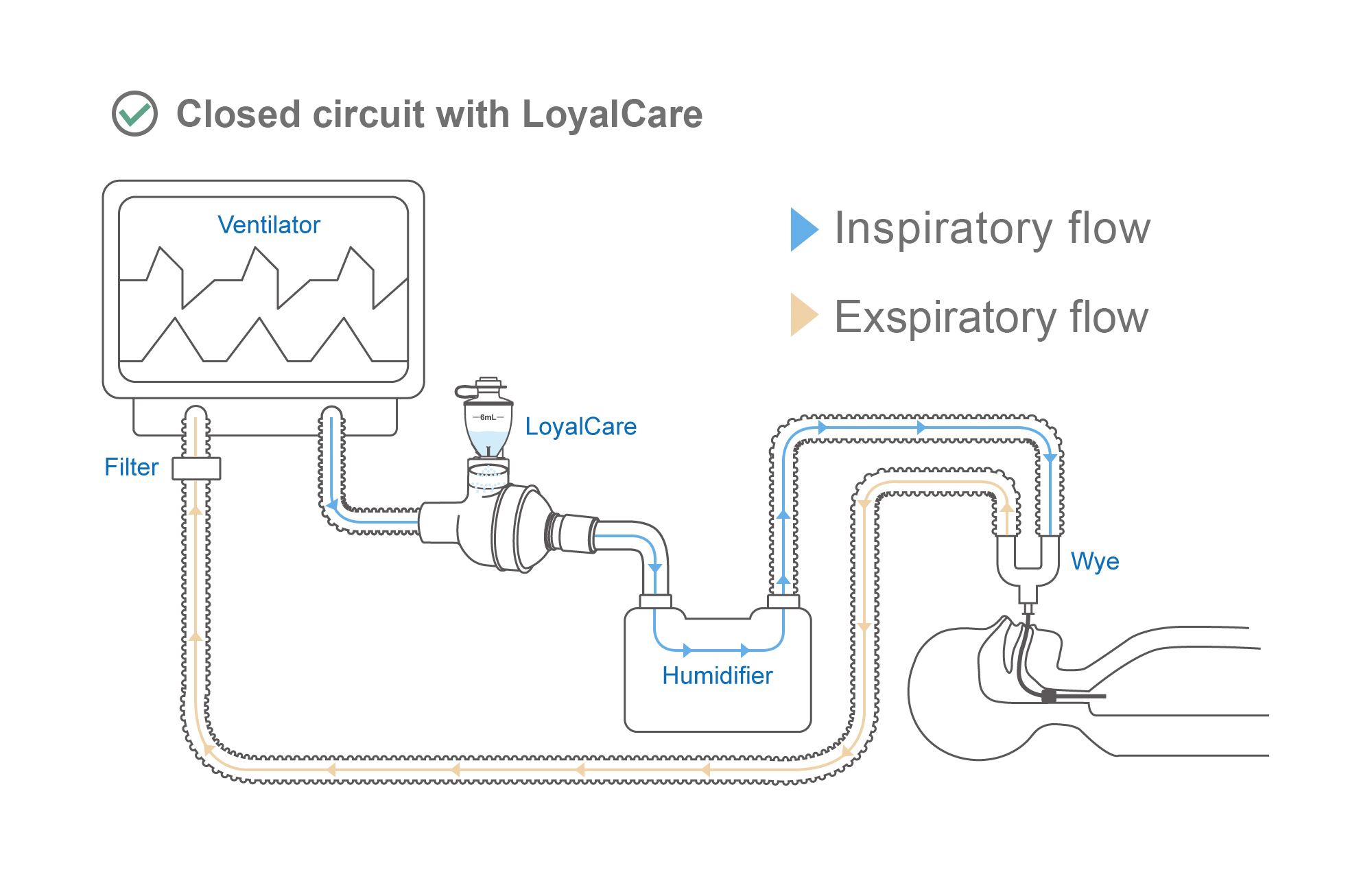
Effectively delivers aerosolized medications to patients during Invasive Mechanical Ventilation (IMV).
NGI (Next Generation Impactor) In-Vitro Performance Data
Non-Invasive Ventilation
Directly compatible with CPAP (Continuous Positive Airway Pressure) and BiPAP (Bilevel Positive Airway Pressure). Can be integrated without supplementary flow or interrupting the therapy cycle.
High-Flow
Allows in-line drug administration without treatment interruption. Maintaining a closed respiratory circuit effectively reduces infection risks and improves therapeutic efficiency.
NGI (Next Generation Impactor) Performance Data
Field and Medical-Grade Design
USB Controller

- ✓ USB-compatible for power bank usage in field operations.
- ✓ Automatically power off within 30 minutes.
- ✓ Dual mounting options for various hospital settings.
- ✓ Standby indicator for clear shift handover without re-confirmation.
T-Piece

- ✓ PP material, heat-resistant and BPA-free for patient safety.
- ✓ Spherical T-Piece increases the amount of drugs directly to the lungs.
- ✓ Compatible with humidifier and heater for comfort and absorption.
- ✓ Can be connected by accessories meets ISO 5356 standard.
Elevating Clinical Care
- ● Single patient use.
- ● Using time: 28 days (4 treatments per day).
- ● Intended user : Neonates (29 days or older) and adult patients.

✓Protecting Healthcare Workers’Safety
The closed-circuit design minimizes the risk of exposure to pathogens.
✓ Precise Drug Delivery Control
Effective drug utilization through low residue and high inhalation efficiency.
✓Reducing Healthcare Burden
Compared to metered-dose inhalers (MDIs), ICU length of stay is reduced by 28%. Compared to jet nebulizers, exposure is reduced by 14%.¹
1. Source: CADTH Health Technology Review (2021)
Streamlined Operation

① Connect the AC/DC power adapter to the power outlet.

② Connect the USB controller to the power adapter.

③ Check the blue light on the USB controller is on.

④ Plug the nebulizer cable to the nebulizer.
| Model | MB05T01 |
| Method of operation | Active Vibrating Mesh Technology |
| Power Supply | AC/DC Adapter: - Please use a power adapter that meets the requirements of IEC60601-1 and CCC certification standards. - Specification: 5V DC, 1A |
| Vibration Freq. | Approx. 120 (kHz) ±10% |
| Nebulization Rate | ≥ 0.2 mL / min |
| Particle Size | MMAD ≤ 5μm |
| Capacity of Nebulizer | Maximum 6 mL |
| Intended user | 29 days Neonatal or older and adult. Operator must be legally practicing medical personnel. |
| Operating Env. | Temp: 10°C - 40°C; RH: 30 - 85%; Pressure: 800 - 1060hPa |
| Storage Env. | Temp: -20 - 70°C; RH: 20 - 75%; Pressure: 800 - 1060hPa |
| Service Life | 28 days (4 treatments per day). |
| Weight | USB Controller: Approx. 100g Medication Cup: Approx. 6.5g |
| Kit Contents | USB Controller, T-Piece, Nebulizer and Adapter. |